Since the outbreak of the global pandemic, we as society have been reminded that health is the most important good in life. In the absence of health, a virus such as COVID-19 can cause the world to stand still for months. Alongside health, hope is important to survive. Hope is what keeps us alive and motivates us to continue – no matter how difficult the circumstances are. With vaccines for COVID-19 already being administered, cold chain logistics for pharma products is in the spotlight.
Vaccines require a resilient cold chain with specific requirements for quality assurance. These products can save lives. As their key handlers, logistics companies can also save lives and their responsibilities go beyond transporting boxes via land, air or ocean from A to B. In addition, knowing how to ensure a stable and reliable cold chain for certain products becomes key.

Logistics for pharmaceuticals are demanding, complex, and different to any other industry. For biopharma products, a reliable temperature-controlled supply chain is essential to save lives. A cold chain is defined as a supply chain with cargo that requires end-to-end temperature control throughout transit. An unbroken cold chain is a sequence of storage efforts that maintain a given temperature range (e.g. 2⁰-8 ⁰C) to transport a sensitive product, including pharmaceutical drugs and biological materials. If the cold chain is broken, the product can become damaged, causing it to lose integrity or, in the worst case, harm the patient. As summarized by Veronique Dameme, Head of Global Vertical Market Healthcare at DB Schenker: “You have to keep in mind, that at the end of the supply chain there is a human being!”.
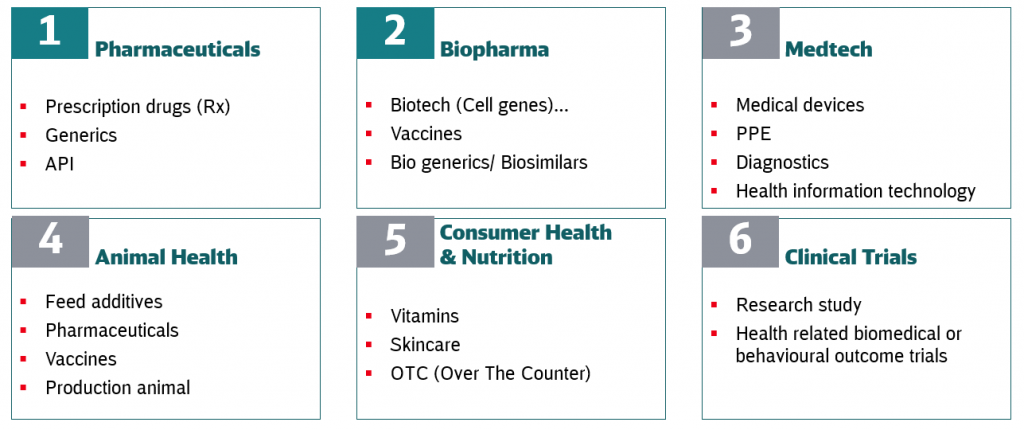
The healthcare business is a steadily growing market for logistics service providers – especially when it comes to temperature-controlled logistics. Generally speaking, the healthcare business is divided into six segments. When it comes to cold chain logistics for pharma products, the Biopharma segment is both the most interesting and most challenging area – particularly when the latest product developments and treatments are considered.
Market development in cold chain transport for pharmaceuticals
The demand for healthcare products is growing, resulting in increased opportunities for logistics service providers to offer solutions in cold chain logistics for pharma products. Market research experts from Grand View Research estimated that the global pharmaceutical logistics market size is expected to grow by a compound annual growth rate of 7.3 percent from 2020 to 2027. In 2019, the market size was valued at USD 69 billion. The short-term impact of COVID-19 on the market is the crucial factor for pharmaceutical logistics’ fast-paced growth across the globe.
Key influences and aging populations are growing around the world. Under these circumstances, the demand for more personalized healthcare rises. The COVID-19 pandemic has also, of course, increased the market growth.

Pharmaceutical products are often transported via air freight. In 2018, the International Air Transport Association (IATA) estimated a 6 percent increase in cold chain logistics for pharma by 2021, reaching USD 396 billion in global biopharma sales. Five key drivers were identified: first, the continuing transition to biologically based products; second, the tightening requirements for life sciences shipments; third, the growing internationalization of pharmaceutical trade; fourth, the rising introduction of biosimilars; and fifth, the broader adoption in underdeveloped economies. COVID-19 vaccine transport was not included in this study, which could now be evaluated as the sixth driver.
Cooling and safety requirements for pharmaceuticals
To set the global standard in pharma distribution, two guidelines have been developed: Good Distribution Practice (GDP) from the European Union and Center of Excellence for Independent Validators (CEIV) by IATA. GDP assists wholesale distributors in procuring, holding, supplying, or exporting medicinal products, while CEIV focuses on air freight and temporary storage.
The CEIV has defined four key areas to ensure the integrity of temperature-sensitive products: preventing sanitary issues caused by temperature excursions during transportation, improving the handling of pharmaceutical products and compliance with existing regulations and standards, elevating staff competency through an efficient and robust training program, and creating a global and consistent certification that industry can rely on.
Cold chain logistics for pharma or other healthcare products is completely different to the transport of perishables. The temperature standards for healthcare products as defined by the World Health Organization (WHO) are as follows: deep-frozen (-20⁰C to -70⁰C), frozen (-20⁰C), sensitive but not frozen (2⁰ to 8⁰C), cool (8⁰ to 15⁰C) and room temperature (15⁰ to 25⁰C).
Transport and packaging of pharmaceutical products with cold chain requirements
COVID-19 has severely affected global supply chains. Never before has the logistics industry and transport routes been so disrupted. The breakdown of international long-haul passenger flights and related wide-body belly capacity has led to massive shortages in air freight capacity when pharmaceutical products have been needed the most. In the pharma industry, logistical delays are not an option.
For biopharma products such as vaccines and other temperature-controlled goods, packaging solutions are just as important as transportation. Accordingly, logistics companies and airlines specializing in pharma transportation must offer services that match the higher demands. There are three types of packaging solutions for temperature-controlled transports: active, hybrid, or passive containers.
Active containers have active temperature control, enabling containers to stay within a defined temperature range. While active containers have electrical cooling and heating systems or cooling with dry ice, hybrid solutions are a combination of enhanced technology and systems to maintain a certain temperature. Passive containers are insulated boxes or containers without any active temperature control unit, for example a fan or electrical power unit.
For temperature-sensitive products, active cold chain packaging solutions are the safest choice. Temperatures are adjusted according to ambient temperatures with broad ranges, meaning active containers can be plugged in to cool in case of delays. Recently, however, hybrid containers have made a strong move into the market, leading to changes in the market share.
Economically, the choice must be analyzed in detail. Active container costs look higher at first glance, but take up much less cargo space and require less handling. Considering all cost factors, results can be different in terms of Total Landed Costs. Large active units often have a lower Total Landed Cost compared to passive solutions.

The price of air freight has risen, meaning some pharmaceutical companies are looking for alternative transport for their life science products to reduce costs. For products with a lower sensitivity on their stability data, ocean freight can be a viable option. Again, the right combination of transport, packaging, and monitoring is necessary to secure the product quality at the destination.
“In times like these, we are facing a situation where customers are searching for a logistics service provider who acts as a partner for their business. To secure the integrity of a temperature sensitive product, it is important to rely on a partner who knows, understands and is able to manage – especially in times of uncertainty – shipments with the right care!”
For Christoph Hemmann, the top five success factors for logistics service providers are:
- Quality management and service quality must be part of the DNA for a logistics service provider
- Having a global network and highly skilled and trained staff is critical for the healthcare business
- Offering the best temperature-controlled packaging solutions (active, hybrid, and passive)
- Centralized monitoring of sensitive products and shipments
- Reliable performance with smooth processes and capacity solutions to minimize risks
Cold chain logistics for pharma products
With everyone along the pharmaceutical supply chain breaking new ground, it is best to use a logistics service provider who understands and lives its business on a global scale. DB Schenker, for example, immediately establised new transport routes to make up for disruptions during the pandemic. The logistics service provider recently set up new routes with Cargolux to support increased demand for pharma shipments, and a dedicated reefer department has been established to offer active containers to customers.
To ensure that all goods receive the optimal mix of transport elements, DB Schenker has developed a new healthcare solution: DB SCHENKERlife+. Customers can now add transport and additional services to their product requirements, including charter capacities such as unique GDP-compliant flights, Connect2Track options, and 24/7 monitoring. Packaging supply and sourcing can be added on top, alongside additional Internet of Things devices. DB Schenker offers GDP-compliant processes and has subject matter experts on hand to advise and consult customers in finding the right service mix.
Health and hope are the main drivers for every human to live. COVID-19 vaccine transport is currently a challenge for logistics providers worldwide. In this moment, the provision of secure and reliable logistics services is a question of honor which has never felt more important to society.
Published: February 2021